For emergency physicians, intracranial hemorrhage (ICH) is always at the top of the differential when a patient presents with altered mental status.
Although uncommon, the relative risk of ICH during pregnancy and up to five weeks postpartum is 5.6 times higher than in nonpregnant women and carries an in-hospital mortality rate of 20%.1,2
We present a case of a woman of advanced maternal age who presented with altered mental status at 10 days postpartum. She was subsequently diagnosed with intracranial hemorrhage secondary to both preeclampsia and HELLP (hemolysis, elevated liver enzymes, and low platelets) syndrome.
Case Report
The previously healthy 44-year-old Black patient, G5P4014, presented to the ED for altered mental status 10 days postpartum from a spontaneous vaginal delivery. According to her partner, she began experiencing a sudden onset headache about an hour prior to arrival. The partner denied any triggers, including recent trauma. Prior medical records from her last delivery indicated that the patient had an unremarkable prenatal course and an uncomplicated vaginal delivery. Her records did not indicate any antihypertensive or anticoagulant use.
Triage vitals were significant for a blood pressure of 202/113 mmHg, respiratory rate of 30 bpm, heart rate of 44 bpm, rectal temperature 36.8 degrees Celsius, and oxygen saturation 99% on room air. On initial examination, the patient was somnolent, eyes opened to command, withdrew from pain, no comprehensible speech. There were no obvious signs of trauma.
Given the vital signs and clinical presentation, the patient was sent immediately to CT imaging to evaluate for intracranial injury. Initial imaging (Figure 1) revealed bilateral frontal intraparenchymal hematomas, small bilateral frontal subarachnoid hemorrhages, and moderate right and mild left intraventricular hemorrhage in the lateral ventricles. Laboratory workup revealed: WBC 8.0 K/mm3; Hgb 8.4 g/dL; Hct 23.2%; Plt 70 K/mm3; AST 95 IU/L; ALT 55 IU/L; T. Bili 1.8 mg/dl; and LDH 904 IU/L. Coagulation studies were as followed: aPTT 27.4 and PT/INR 22.7/1.3. Urinalysis revealed 3+ proteinuria.
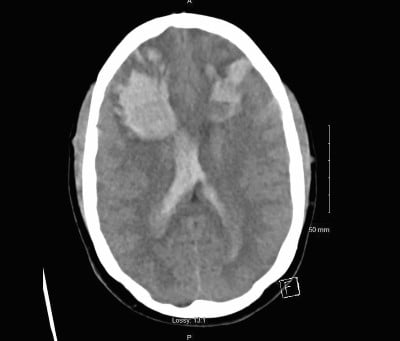
Figure 1
After imaging, the patient became increasingly altered and minimally responsive to pain, requiring intubation for airway protection. Her blood pressure continued to remain elevated, with systolic blood pressures in the 190s mmHg. Propofol, midazolam, magnesium, and nicardipine infusions were started, with improvement in blood pressure to systolic blood pressure to the 140s mmHg. Additionally, the patient was given levetiracetam for seizure prophylaxis and mannitol to reduce intracranial pressure.
Neurosurgery was consulted at a nearby tertiary care center. Our patient was subsequently transferred and upon arrival had emergent bifrontal craniectomy for evacuation of hematoma and bilateral external ventricular drain placement.
After neurosurgical intervention, the patient continued to require ICU level care. On hospital day 10, the patient aspirated and was subsequently diagnosed with right-sided aspiration pneumonia. During this time, she became septic, required vasopressor support, and was started on piperacillin-tazobactam. On hospital day 16, she underwent tracheostomy and PEG tube placement. On hospital day 17, sedation was weaned off, but the patient had little improvement in neurocognitive status. On hospital day 30, the patient was transferred to a long-term care facility for continued ventilatory weaning and monitoring of neurological status. At the time of article submission, she remained at a similar neurological status: able to open eyes to voice but unable to move extremities.
Discussion
ICH is a rare pregnancy complication but an important cause of maternal mortality and is greatest up to 12 weeks postpartum.2 Significant risk factors for pregnancy-related ICH include advanced maternal age, Black race, preeclampsia, and coagulopathy such as HELLP syndrome.3,4,5
Preeclampsia is characterized by hypertension, proteinuria, and edema after 20 weeks gestation.6 The incidence of preeclampsia in the United States is approximately 5% of singleton pregnancies.7 Postpartum preeclampsia can be defined as signs and symptoms of the disease more than 2 days, but fewer than 6 weeks, after birth.8 In a retrospective study, 63% of patients who had late onset postpartum preeclampsia had no diagnosis of hypertensive disease in their current pregnancy, similar to our patient.8 The presenting symptoms are commonly nonspecific and require a high clinical suspicion. Symptoms include persistent or severe headache, visual disturbances, abdominal pain, altered mental status, and new onset shortness of breath.9
HELLP syndrome is characterized by hemolysis, elevated liver enzymes, and low platelets.10 Although it may be a form of preeclampsia, it can present as a separate pathology given that up to 20% of patients with HELLP do not have hypertension or proteinuria.11 The pathophysiology of HELLP is multifactorial and has similar risk factors to preeclampsia.11
Case Management
Our initial differential for our patient included infectious etiologies such as meningitis or encephalitis, hemorrhagic or ischemic stroke, cerebral venous thrombosis, status epilepticus, and carotid artery dissection. As our patient was 10 days postpartum, preeclampsia with severe features became the more likely diagnosis.
Our patient had an unremarkable prenatal course, but she met multiple risk factors for developing both preeclampsia and HELLP syndrome, including advanced maternal age and Black race. Based on her presentation and laboratory findings, we suspected she developed postpartum preeclampsia, which made her more susceptible to ICH because of her acquired coagulopathy from HELLP syndrome. Unfortunately, our patient’s initial triage vitals demonstrated Cushing’s triad: bradycardia, hypertension, and irregular respirations as a result of increased intracranial pressure due to significant intracranial hemorrhage.
Women who experience pregnancy-related ICH are 85 times more likely to die compared to women who do not experience pregnancy-related ICH.2 The goal of ICH management is early recognition to prevent neurological decline and long-term neurological deficits. Management in the emergency department includes immediate head imaging, rapid reduction in blood pressure, and frequent neurological assessments to monitor mental status and airway protection. Ultimately, management requires multidisciplinary collaboration with neurology, neurosurgery, and intensive care, which may mean transferring the patient to an appropriate care facility.
Our case report supports the importance of maintaining a high clinical suspicion for intracranial hemorrhage when a postpartum female presents to the emergency department with acute neurological symptoms. Prompt recognition and management are cornerstones for the patient’s best neurological outcome.
References
1.Kittner SJ, Stern BJ, Feeser BR, et al. Pregnancy and the risk of stroke. N Engl J Med. 1996;335(11):768-774. doi:10.1056/NEJM199609123351102
2. Meeks JR, Bambhroliya AB, Alex KM, et al. Association of Primary Intracerebral Hemorrhage With Pregnancy and the Postpartum Period. JAMA Netw Open. 2020;3(4):e202769. Published 2020 Apr 1. doi:10.1001/jamanetworkopen.2020.2769
3. Bateman BT, Schumacher HC, Bushnell CD, et al. Intracerebral hemorrhage in pregnancy: frequency, risk factors, and outcome [published correction appears in Neurology. 2007 Apr 3;68(14):1165]. Neurology. 2006;67(3):424-429. doi:10.1212/01.wnl.0000228277.84760.a2
4. Toossi S, Moheet AM. Intracerebral Hemorrhage in Women: A Review with Special Attention to Pregnancy and the Post-Partum Period. Neurocrit Care. 2019;31(2):390-398. doi:10.1007/s12028-018-0571-3
5. James AH, Bushnell CD, Jamison MG, Myers ER. Incidence and risk factors for stroke in pregnancy and the puerperium. Obstet Gynecol. 2005;106(3):509-516. doi:10.1097/01.AOG.0000172428.78411.b0
6. Gestational Hypertension and Preeclampsia: ACOG Practice Bulletin, Number 222. Obstet Gynecol. 2020;135(6):e237-e260. doi:10.1097/AOG.0000000000003891
7. Hutcheon JA, Lisonkova S, Joseph KS. Epidemiology of pre-eclampsia and the other hypertensive disorders of pregnancy. Best Pract Res Clin Obstet Gynaecol. 2011;25(4):391-403. doi:10.1016/j.bpobgyn.2011.01.006
8. Al-Safi Z, Imudia AN, Filetti LC, Hobson DT, Bahado-Singh RO, Awonuga AO. Delayed postpartum preeclampsia and eclampsia: demographics, clinical course, and complications. Obstet Gynecol. 2011;118(5):1102-1107. doi:10.1097/AOG.0b013e318231934c
9. Sibai BM. Diagnosis, controversies, and management of the syndrome of hemolysis, elevated liver enzymes, and low platelet count. Obstet Gynecol. 2004;103(5 Pt 1):981-991. doi:10.1097/01.AOG.0000126245.35811.2a
10. Wallace K, Harris S, Addison A, Bean C. HELLP Syndrome: Pathophysiology and Current Therapies. Curr Pharm Biotechnol. 2018;19(10):816-826. doi:10.2174/1389201019666180712115215
11. Fitzpatrick KE, Hinshaw K, Kurinczuk JJ, Knight M. Risk factors, management, and outcomes of hemolysis, elevated liver enzymes, and low platelets syndrome and elevated liver enzymes, low platelets syndrome. Obstet Gynecol. 2014;123(3):618-627. doi:10.1097/AOG.0000000000000140



