A 69-year-old female presented to the emergency department for altered mental status and respiratory distress.
Approximately one month prior, she had been struck by a vehicle and admitted for multiple traumatic injuries including: multiple left-sided subarachnoid hemorrhages, open mandible fracture, left Lefort I fracture, right Lefort II fracture, inferior orbital fracture, left iliac wing fracture, left tibia and fibula fracture, right knee full thickness tear of the anterior cruciate ligament, posterior cruciate ligament, lateral collateral ligament, and biceps femoris tendon. The patient had undergone multiple surgeries for the orthopedic injuries, and her hospital course was complicated by new onset atrial fibrillation. She was discharged from the hospital to a rehabilitation center four days before this presentation.
The rehabilitation facility called EMS because staff found her with right-sided paralysis, altered mental status, and saturating at 80% on room air. She was at her baseline and was working with physical therapy four hours earlier.
Initial vital signs at 15:05 in the ED reveal tachypnea to 28 breaths per minute, oxygen saturation of 91% on 15L supplemental oxygen, BP 130/85 mmHg, and heart rate 80 bpm. She had spontaneous respirations and lung sounds were clear bilaterally. Her initial GCS was 7 (E2, V2, M4). She had a leftward gaze deviation and flaccid paralysis of the right upper extremity; no response to painful stimuli in the right lower extremity. She had a knee immobilizer on the right lower extremity and long leg splint on the left lower extremity with swelling in bilateral lower extremities. Capillary refill in her feet was fewer than 2 seconds. The patient had undergone maxillomandibular fixation; she had a healing scar to her anterior mandible and multiple healing contusions to her face.
A non-contrast CT of the brain showed evidence of an occlusion of the large middle cerebral artery (MCA) stroke. CTA of the head and neck as well as CT perfusion studies were subsequently obtained to further evaluate the stroke. CTA of the head and neck showed hypoattenuation of the left intracranial internal carotid artery (ICA) and MCA M1 segment, highly concerning for acute thromboembolic disease. The CT perfusion also showed large acute left MCA infarction. CTA of the chest showed a saddle pulmonary embolism (PE) with evidence of right heart strain. Her initial high-sensitivity cardiac troponin was significantly elevated to 363 ng/L.
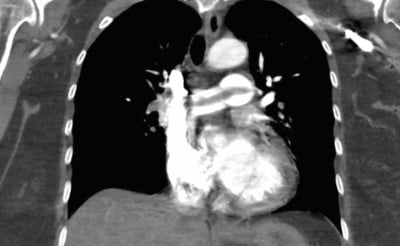
Image 1: Coronal view, CTA chest demonstrating saddle pulmonary embolism
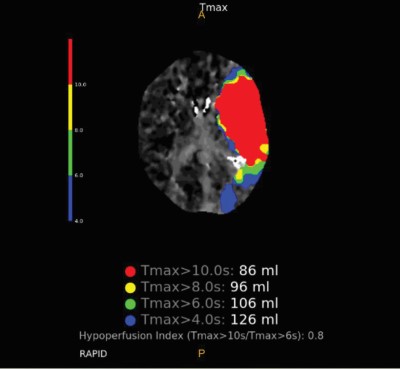
Image 2: CT Perfusion demonstrating significant hypoperfusion to the left MCA territory
Management Considerations
There are few reported cases of concomitant acute ischemic stroke and pulmonary embolism. Given the rare presentation, there are no guidelines on how to manage such a situation. This case was further complicated by the fact that it was a submassive saddle PE with right heart strain and a difficult airway, in addition to the large ischemic stroke.
Recent intracranial hemorrhage was an absolute contraindication for alteplase administration and thus narrowed down the management choices. The remaining considerations included systemic anticoagulation, catheter-directed arterial thrombolysis, endovascular embolectomy, and IVC filter placement. Neurology recommended against systemic anticoagulation secondary to ischemic lesions with high risk of conversion to hemorrhagic stroke over the next few days.
The best course of action at this time was to have interventional radiology (IR) attempt endovascular thrombectomy and embolectomy. Regardless of the outcome of the IR procedures, she would ultimately require an IVC filter for long-term prevention against recurrent emboli.
Airway Considerations
Interventional radiology was consulted and agreed with the plan for ICA and MCA thrombectomy, followed by PE embolectomy. The patient was at high risk of deterioration and needed a definitive airway. Vital signs remained stable with no evidence of obstructive shock from the saddle pulmonary embolism.
Intubating a patient with right heart strain from pulmonary artery clot burden can trigger a downward spiral of cardiovascular collapse. The right ventricle (RV) outlet obstruction causes decreased RV output, which leads to decreased cardiac output from the left ventricle and decreased coronary perfusion. This can lead to cardiovascular collapse. Positive pressure ventilation increases intrathoracic pressure and can further potentiate the RV failure. The sedation may also lead to hypotension and further decreases in cardiac output.
In addition to the very high risk of cardiovascular collapse, this patient also had an anatomically complex airway that further complicated the situation.
Case Timeline
Despite removal of the maxillomandibular fixation wires, the patient was not able to open her mouth greater than 1 centimeter. A cricothyrotomy kit was brought to the bedside in case of rapid decompensation. The anesthesiologist came to evaluate the patient for potential nasotracheal intubation and agreed that her recent multiple facial fractures made her anatomy for orotracheal or nasotracheal intubation nearly impossible. Her oxygen saturation remained in the low to mid-90s on 15L non-rebreather.
Day 1, 18:43: At this point, the neurointerventionalist did not want to further delay the ICA and MCA thrombectomy; thus the patient was brought to the interventional suite on the non-rebreather. Unfortunately, the IR team was unable to retrieve the thrombus.
19:45: Immediately after the thrombectomy attempt, the patient was brought to the OR with ENT for an urgent awake tracheostomy and subsequently brought to the ICU.
22:20: The patient returned to the IR suite for an embolectomy. At the beginning of the procedure, she desaturated and became hypotensive, leading to a PEA cardiopulmonary arrest. Return of spontaneous circulation was achieved after one dose of epinephrine and one round of chest compressions. She returned to the ICU for hemodynamic stabilization.
Day 2: The patient decompensated and was in obstructive shock with vasopressor support. Embolectomy was attempted again after hemodynamic stabilization. IR successfully decreased the clot burden and placed an IVC filter.
The patient’s hemodynamics and mental status gradually improved and after approximately one month in the hospital, she was able to respond to commands and nod in response to questions. She had residual hemiplegia and aphasia, but was ultimately discharged to a long-term care facility with a tracheostomy and PEG tube.
Take-Home Points
- Concurrent acute ischemic stroke and submassive PE is a rare occurrence, and there is no sufficient evidence to support a particular method of management. It is important to weigh the benefits and risks of the various options for each patient presentation. The most common options based on literature review are IV thrombolysis, mechanical thrombectomy, or catheter directed thrombolysis. Some case reports recommend heparin after thrombolysis; however, our patient was at too high of risk for hemorrhagic conversion of the ischemic stroke.
- Positive pressure ventilation can worsen cardiac output and cause cardiovascular collapse in cases of right heart strain from pulmonary embolism. Consider embolectomy to help improve oxygenation prior to intubation when possible.
- If possible, utilize hospital resources (such as anesthesiology, surgery, or ENT) to help manage a difficult airway before resorting to emergent cricothyrotomy.
References
Bryce YC, Perez-Johnston R, Bryce EB, Homayoon B, Santos-Martin EG. Pathophysiology of right ventricular failure in acute pulmonary embolism and chronic thromboembolic pulmonary hypertension: a pictorial essay for the interventional radiologist. Insights Imaging. 2019 Feb 13;10(1):18.
Farkas J. Eight pearls for the crashing patient with massive PE. EMCrit Project. PulmCrit (EMCrit). Published June 4, 2017. Accessed Oct. 16, 2022.
Saleh Velez FG, Ortiz Garcia JG. Management dilemmas in acute ischemic stroke and concomitant acute pulmonary embolism: Case series and literature review. eNeurologicalSci. 2021 Apr 15;23:100341.