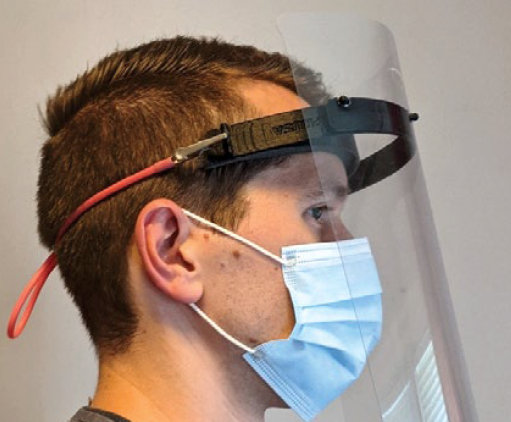
Supply chain disruption during the COVID-19 pandemic has forced healthcare systems to look for alternative ways to meet dramatically increased demand for personal protective equipment (PPE).
One potential solution to this problem is 3D printing of PPE. However, relatively few of these 3D-printed PPE designs have been evaluated by healthcare professionals, and many require extensive redesign to make them practical and safe for use. This paper outlines an interdisciplinary approach to evaluating and refining a 3D-printed face shield design for use in the Emergency Department of a level 1 Trauma center in eastern North Carolina. Survey data indicated that users of the 3D-printed face shields considered them equal to disposable shields in comfort and usability, and superior to disposable shields in durability and protection. A majority indicated they preferred using the 3D-printed shield. Given these results, we believe that 3D printing of PPE is a viable approach to mitigating future PPE supply chain disruptions.
Introduction
During the early days of the COVID-19 pandemic in 2020, health care systems across the United States and worldwide experienced a dramatic disruption to supply chains providing PPE. Hospitals were forced to sanitize and reuse PPE and to look for alternative ways to meet the increase in demand. At Vidant Medical Center in Greenville, NC, one of those creative solutions was to manufacture PPE using 3D printers.
While 3D printing companies and enthusiasts around the country were actively designing and printing PPE such as face shields and N95-type respirators for personal use, few were being used and evaluated by healthcare professionals. In the months since the beginning of this project, several papers have been published describing similar efforts to produce 3D-printed PPE around the world1,2,3. To meet our own local needs at Vidant Medical Center in eastern North Carolina, a group of ECU students decided to create a 3D-printed face shield that would be suitable for use in the clinical setting. After several rounds of testing and refinement with a focus group of trauma surgeons and ED physicians, the final product was distributed to Vidant Medical Center ED physicians and survey data were collected to evaluate the face shields on several qualities including durability, safety, usability, and user preference.
This project was a highly interdisciplinary undertaking. Brody School of Medicine students directed the project with support from local trauma surgeons and ED physicians. ECU’s School of Art and Design faculty and students aided in printing and design. A local marine supply company graciously donated their CNC machine for cutting plastic to specifications. ECU’s School of Dental Medicine donated silicon dental bib clips to use as headbands. We are grateful for their timely help to keep our frontline workers safe during the early days of the COVID-19 pandemic.
Materials and Methods
Face shields were assembled from a 3D-printed visor and a few widely available commercial products. These products include a silicon dental bib clip, weatherproofing window foam strips, and clear plastic sheeting.
For a headband, the original design from PRUSA4 recommended the use of elastic buttonhole fabric, but given supply chain issues with fabric in the early days of the pandemic we sought alternative options. Instead, silicon dental bib clips served as effective headbands. Their alligator clips attached easily to the visor, their length was adjustable, and they were easy to clean after use. These clips have an estimated cost of $1.50 per unit.
Weatherproofing window foam strips served as a cushion for the user’s forehead. Following testing of several different brands, Duck Brand Foam Weatherstrip Seal was chosen because the adhesive was the best for attachment to the printed visor. This cushion cost $0.35 per face shield.
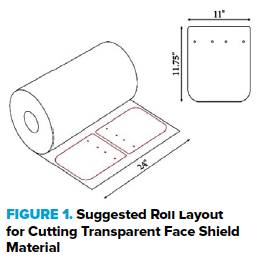
Whereas the bib clip and foam strips require minimal processing, the clear plastic sheeting must be cut into 11” x 11.75” rectangles with rounded corners and holes punched into the top to attach to the visor. These dimensions were chosen to maximize protection without compromising comfort and head mobility. A local marine supply group provided a CNC machine capable of cutting a large number of shields from a roll of plastic. A diagram of the correct dimensions and peg hole locations is shown in Figure 1 below. The plastic sheets we used cost $0.64 per face shield.
To 3D print the visor component using PLA material, Makerbot 3D Replicator + and Makerbot 3D Replicator 5th Generation printers were used. After considering several publicly available designs, the PRUSA CR3 design was chosen for its robustness and stability, although this came at the cost of increased materials and printing times. This template was then modified with the help of ECU School of Design students using Maya 3D editing software. In its final iteration, the plastic cost $1.48 per visor. To maximize printing efficiency, a stacked design was used so that a single print run produces 4 visors, as seen in Figure 2.
A Makerbot Replicator+ printer can produce 4 shields in 8 hours. The Innovation and Design Laboratory at ECU has a fleet of over 20 Makerbot Replicator+ and Replicator 5th generation printers (Figure 3 below), making it capable of producing 120 visors per day if all printers are used to maximal capacity. However, in practice, the printers experience malfunctions on approximately 10-20% of printing runs, necessitating some level of supervision to maintain maximal production.


The completed product currently has a cost of $3.97 per face shield, not including the cost of the printer or electricity. Although this is likely a greater cost than most disposable face shields used by hospitals, this shield is designed to be sanitized and reused many times. The final product is shown in Figure 4 below.
Figure 4. Final face shield product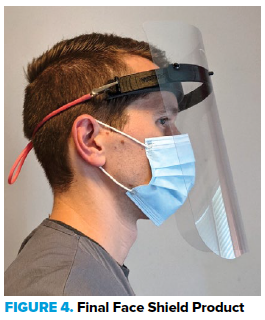
The completed face shield was distributed to residents and attending physicians in the ED of Vidant Medical Center. A survey was then distributed to users of the face shields to evaluate various qualities of the shield compared to the hospital’s standard disposable shield (McKesson product #1049043).
Numerical rating scales from -50 to 50 were used to compare the shields based on certain attributes, where -50 represented a complete preference for disposable shields, 0 represented no preference, and 50 represented complete preference for the 3D-printed shield. Survey questions asked users to compare durability, protection, ease of communication, ease of performing procedures, comfort, and overall preference between the 3D-printed shield and disposable shield. Additional questions included the total hours of use, ease of cleaning, and whether corrective lenses were worn with the shield.
Results
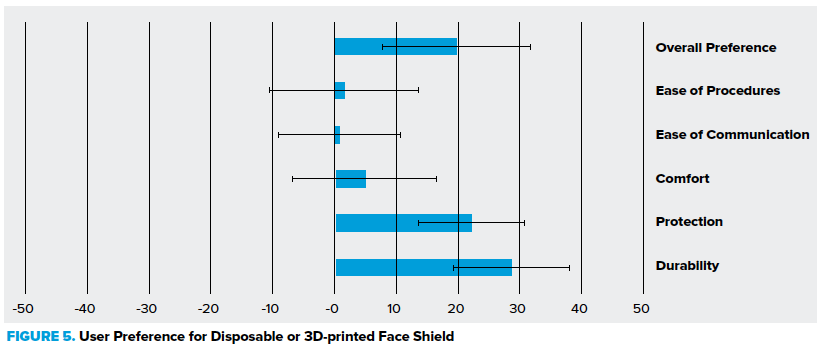
A total of 18 survey responses were submitted. Respondents used the shields for an average of 41 hours each. A graphic representation of the data comparing disposable and 3D-printed face shields is shown in Figure 5.
Figure 5. Survey responses
This chart summarizes the distribution of survey responses comparing qualities of the 3D-printed shield and the standard disposable shield. A score of 50 indicates complete preference for the 3D-printed shield with respect to that quality, -50 indicates complete preference for the disposable, and 0 indicates no preference. Error bars represent 95% CI.
Respondents rated the ease of sanitizing the face shield favorably with an average rating of 15.71. For sanitizing, it was recommended to use ethanol wipes or spray as ethanol does not degrade the PLA material or the acetate sheeting used. PLA material is heat-labile and does not withstand autoclaving, and bleach also degrades the PLA material over time.1
Respondents also indicated they would be highly likely to use the shields in the event of a future PPE shortage with an average rating of 36.5, where a rating of -50 represents not likely at all and 50 represents very likely.
Discussion
Durability: 3D-printed shields received a favorable durability rating of 28.61. This product is designed to be reused over multiple shifts. Printing settings can be altered to increase the density and strength of the visor even more, but this increases weight, materials, and printing times.
Protection: 3D-printed shields offered greater protection compared to the disposable shields as perceived by respondents, with an average rating of 22.25, likely due to its larger plastic shield.
Comfort: Respondents indicated that the 3D-printed shield was roughly equal to the disposable in comfort with a rating of 4.82. Both have a foam pad to reduce pressure on the forehead. However, the 3D-printed shield is heavier and its strap thinner, placing more pressure on the back of the head. Future iterations may implement a wider strap for improved comfort.
Ease of communication and performing procedures: Respondents reported that the 3D-printed shield was equal to the disposable in both aspects.
Overall preference: The 3D-printed shield was overall preferred with a rating of 19.81. This may be due to user perception of increased protection and durability while retaining similar comfort and usability. Focus group feedback suggested that a major advantage of the 3D-printed shield was increased ventilation through the top visor portion, which reduced fogging.
There are several limitations of this study. The sample size was small and included providers from just one ED. The survey recorded user opinions about the face shields, but no formal evaluation of their effectiveness in reducing droplet transmission was undertaken. Additionally, no formal evaluation of the effectiveness of sanitizing was performed as has been done in other studies.3
Conclusion
This project demonstrated the feasibility of supplementing a hospital’s PPE supply through 3D printing techniques. This approach was cost-effective and created a durable, reusable product rated favorably by providers. In the event of future supply chain disruption, 3D printing can supplement the PPE needs of frontline providers. Future research should investigate ways to optimize the design, verify the shield’s efficacy in reducing infectious disease transmission, and to illuminate other uses of 3D printing in healthcare.
References
- Zhang PC, Ahmed Y, Hussein IM, Afenu E, Feasson M, Daud A. Optimization of community-led 3D printing for the production of protective face shields. 3D Print Med. 2020 Nov 23;6(1):35. doi: 10.1186/s41205-020-00089-y. PMID: 33230665; PMCID: PMC7682762.
- Wesemann C, Pieralli S, Fretwurst T, Nold J, Nelson K, Schmelzeisen R, Hellwig E, Spies BC. 3-D Printed Protective Equipment during COVID-19 Pandemic. Materials (Basel). 2020 Apr 24;13(8):1997. doi: 10.3390/ma13081997. PMID: 32344688; PMCID: PMC7215430.
- Mostaghimi A, Antonini MJ, Plana D, Anderson PD, Beller B, Boyer EW, Fannin A, Freake J, Oakley R, Sinha MS, Smith L, Van C, Yang H, Sorger PK, LeBoeuf NR, Yu SH. Rapid prototyping and clinical testing of a reusable face shield for health care workers responding to the COVID-19 pandemic. medRxiv [Preprint]. 2020 Apr 15:2020.04.11.20061960. doi: 10.1101/2020.04.11.20061960. Update in: Med (N Y). 2020 Jun 19; PMID: 32511612; PMCID: PMC7276039.
- Prusa Face Shield stl. https://www.prusaprinters.org/prints/25857-prusa-face-shield. Last accessed 8/4/2021.
- 3Dversktan Protective Visor. https://3dverkstan.se/protective-visor/. Last accessed 8/4/2021.