A 64-year-old male presents to the ED for blood in his urine, which started this morning, followed by inability to urinate spontaneously in the afternoon.
He complains of lower abdominal pain and fullness. He has a history of kidney stones more than 30 years ago, hypertension, diabetes mellitus (type 2), and chronic kidney disease stage 5 (CKD) for which he has been undergoing assessment for renal replacement therapy by outpatient nephrology for the past 4 months. He takes aspirin 81mg daily for CAD prevention; otherwise, he is not on blood thinners. On exam, he is hypertensive with otherwise normal vital signs. He appears uncomfortable, with distention and tenderness of the lower abdomen upon palpation. His genitourinary exam is normal. During the initial assessment, the nurse obtains a urinalysis and culture from a straight in-and-out catheter to drain his bladder, which provides immediate relief of his pain. The urine is grossly bloody upon inspection.
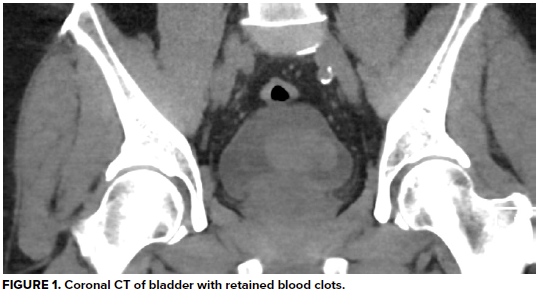
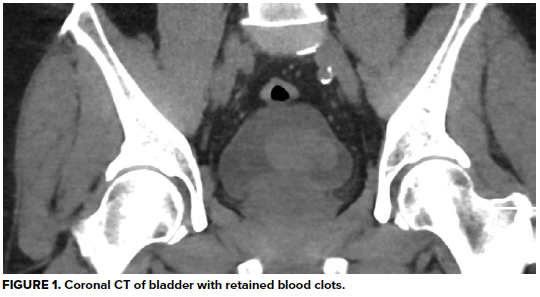
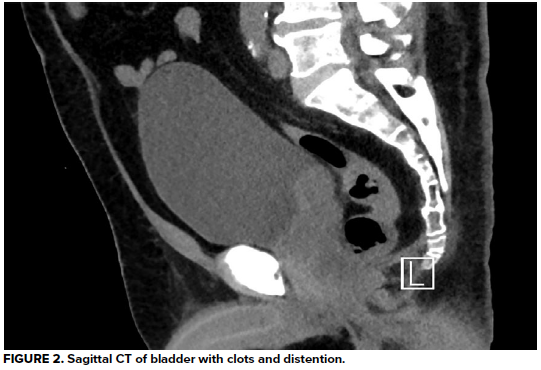
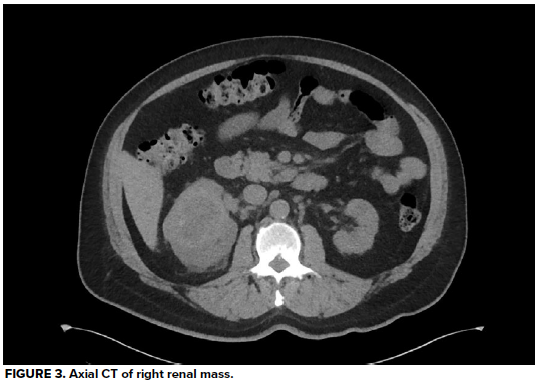
Urinalysis demonstrates 3+ blood and >50 RBC/hpf, but negative leukocyte esterase, negative nitrates, and no bacteria. His hemoglobin is at baseline, as are his BUN and creatinine, consistent with stage 3 CKD. A CT abdomen/pelvis without contrast reveals a large heterogeneous right renal mass with surrounding perinephric stranding, concerning for primary renal malignancy. There is also hyperdense material in a moderately distended bladder most likely reflecting blood clots, as well as scattered periaortic adenopathy which, given the context, raises concern for locally metastatic disease. He is unable to void spontaneously again, even after a fluid bolus.
Introduction
Hematuria is a common complaint among patients presenting to the emergency department. Hematuria is an understandably distressing symptom. Thankfully, the distress experienced by patients with hematuria is often worse than the conditions diagnosed within the ED. Nonetheless, there are several emergent conditions the emergency physician should consider when evaluating a patient with hematuria.
Hematuria is defined by the American Urologic Association (AUA) as greater than three red blood cells (RBCs) visible under a microscope under a high-powered field (hpf) on a standard urinalysis (i.e., greater than 3 RBC/hpf).1 In practice, most labs running urine specimens utilize flow cytometry rather than microscopy to automate the counting of RBCs for standard urinalyses. Hematuria is divided into two types: microscopic hematuria and macroscopic (or gross) hematuria. Microscopic hematuria can only be diagnosed by urinalysis. Macroscopic hematuria is any urine with visible blood or blood-tinged coloring observed with the naked eye. Microscopic hematuria is a common incidental finding in the ED—macroscopic hematuria is more often why a patient presents to the ED for evaluation.
Microscopic Hematuria
There are few immediately life-threatening causes of microscopic hematuria, but the finding itself, especially in isolation, often warrants outpatient evaluation. Common causes of microscopic hematuria include urinary stones, infection, viral illnesses, trauma, vigorous exercise, sexual intercourse, menstruation, renal disease, or recent genitourinary instrumentation.1, 2 Microscopic hematuria in an ED patient experiencing pain or weakness rarely establishes a specific diagnosis by itself, but is often a guidepost in identifying those who will go on to be diagnosed with infection, stone, or even renal failure. Disposition of these patients will depend on the clinical context.
For adult patients with asymptomatic microscopic hematuria, only about 2% will go on to be diagnosed with serious urologic disease such as renal cell cancer, bladder cancer, or prostate cancer.3 In the absence of any other identifiable causes of microscopic hematuria, a patient should receive a referral to their primary care doctor for repeat testing of their urine. Persistent microscopic hematuria on two or more urinalyses when benign causes (such as sexual intercourse or exercise) are excluded warrants an outpatient urology referral for advanced imaging and cystoscopy (especially for patients over 35 years-old or with risk factors for urological malignancies such as smoking, chronic UTIs, occupational exposure to aniline dyes or benzenes). A nephrology referral is additionally recommended if significant proteinuria, cellular casts, or dysmorphic red cells are identified, all of which may suggest a glomerular etiology of hematuria.1, 4
Discharged patients with asymptomatic microscopic hematuria should be referred to their primary care doctor for repeat urinalysis and subsequent referrals to urology and/or nephrology if the microscopic hematuria persists.
Macroscopic Hematuria
Macroscopic (or gross) hematuria is visible to the naked eye. Only 1 ml of blood is required to make a liter of fluid appear grossly hemorrhagic, so even a small amount of blood mixed into the urine can yield alarming results.4 Conversely, the urinary tract can be a source of life-threatening hemorrhage. Initial management of the unstable patient with gross hematuria is, like all critical patients, focused on the ABCs of resuscitation. Unstable patients with gross hematuria should be treated like other patients in hemorrhagic shock with early and emergent hemostatic transfusion. Causes of such life-threatening bleeding include renal tumors, arteriovenous malformations (AVMs), aortocaval fistulas, complicated/septic ureterolithiasis, and trauma, each of which are discussed below.
Emergent Causes of Gross Hematuria
Renal tumors such as renal cell carcinomas (the most common renal cancer) or other malignancies with a predilection for renal metastasis (such as melanomas, lung, breast, gastric, and pancreatic cancers) may present with gross hematuria.5 Such renal tumors with disordered angiogenesis may lead to uncontrolled hemorrhage should a vascular bed rupture into the renal collection system. Along with rapid urological consultation and evaluation, these patients may also benefit from a vascular surgery or interventional radiology consult for trans-arterial embolization of the tumor or even the entire kidney.6, 7 Renal cell carcinoma is primarily treated with nephrectomy, which is usually curative when performed prior to tumor metastasis.5
Renal arteriovenous malformations within the kidney can cause gross hematuria, and may be treated with a vascular or interventional radiology consultation for IR embolization.8
An aortocaval fistula is an abnormal connection between an aneurysmal abdominal aorta and the inferior vena cava (IVC). Increased arterial-level pressures in the low-pressure IVC causes increased venous congestion in the pelvic organs, including along the bladder wall. These bladder wall varices can then rupture, leading to gross hematuria.9 Although rare, aortocaval fistulas are usually detected by CT, and warrant emergent vascular surgery consultation.8
Gross hematuria is present in up to 30% of patients suffering from a kidney stone.10 Nephrolithiasis causing obstruction, especially when signs of infection are present on urinalysis, requires emergent urological evaluation and treatment. Additionally, urine and blood cultures should be obtained promptly and broad-spectrum antibiotics rapidly initiated for the unstable patient presenting with sepsis secondary to an infected kidney stone. Patients with suspected obstruction or infection should receive CT imaging for urological planning purposes, as should patients with suspected first-time kidney stones. CT can also be a helpful tool in considering alternative diagnosis, particularly in the sick or elderly patient presenting with undifferentiated abdominal or flank pain.10 Rapid point of care ultrasound may also be used to determine the laterality of the stone causing obstruction by evaluating for hydroureter and/or hydronephrosis.11 The sensitivity and specificity of ultrasound are 84% and 53% respectively, compared to CT, which has a sensitivity and specificity of 95% and 98% respectively.11
Traumatic causes of hematuria should be evaluated with a retrograde urethrogram to rule out urethral injury, usually followed by CT with IV contrast to evaluate for other injuries.12
Urinary Obstruction
Hemorrhagic debris in the dependent portions of the bladder will eventually clot and obstruct the bladder neck, leading to acute urinary retention. Placement of a large-diameter three-way foley will provide immediate relief to the obstructed patient. Following placement of a three-way catheter, continuous bladder irrigation (CBI) should be initiated to wash out residual clot from the bladder to avoid further obstruction. The decision to initiate CBI is typically done at the recommendation of a urologist. In addition, strictly measuring net urine output in a patient treated for urinary retention is necessary to identify post-obstructive diuresis, a known and potentially fatal complication following relief of urinary obstruction.13 Post-obstructive diuresis is an abnormal amount of urinary output following bladder decompression (usually over 200ml in the first two hours) which can lead to electrolyte derangements, dehydration, hypotension, and even shock.
Non-emergent Causes of Gross Hematuria
Other causes of gross hematuria include urinary tract infection (UTI), uncomplicated nephrolithiasis, urethritis/sexually transmitted infection, schistosomiasis (the most common cause of gross hematuria in underdeveloped countries), and iatrogenic causes.8 A 1987 study demonstrated foley catheterization only induced hematuria in 17% of patients, and even then the amount of hematuria never exceeded 3 RBCs/hpf.14 Conditions which may be mistaken for hematuria by patients include menstruation as well as the ingestion of substances which may turn the urine red such as beets, rhubarb, berries, food colorings, and medications including nitrofurantoin, rifampin, phenazopyridine (commonly sold over-the-counter under the brand names Azo or Pyridium for the treatment of suspected UTI), hydroxychloroquine, and iodine.4 Careful history-taking can help identify these less emergent (or entirely benign) causes, thus guiding efficient management and disposition of the stable patient presenting with hematuria. Laboratory evaluation of hematuria should include complete metabolic panel (CMP) including blood urea nitrogen (BUN) and creatinine, complete blood count (CBC), coagulation studies if the patient is on blood thinners, and of course urinalysis to evaluate for the presence of infection, proteinuria, and casts.
Imaging
The decision to image an otherwise stable patient with hematuria in the ED is made on a case-by-case basis. Interestingly, the American College of Radiology (ACR) Appropriateness Criteria for hematuria states that a CT urogram is “usually appropriate” for any adult patient presenting with gross hematuria.15 The ACR’s criteria does not qualify this recommendation or attempt to sub-classify which patients with hematuria may benefit from advanced imaging more than others. There is no unifying consensus regarding the most appropriate imaging criteria or even the most appropriate modality.16 Most ED physicians choosing to image their patient with hematuria opt for a CT of the abdomen/pelvis with IV contrast to evaluate for a broader array of diagnoses, that is, if they are not suspecting kidney stone, in which case a CT renal protocol without contrast is the study of choice. However, in the otherwise stable patient with gross hematuria in which there is low suspicion for other intra-abdominal pathology (such as an abdominal aortic aneurysm), a CT urogram will provide better resolution of the renal and urothelial tissues to evaluate for malignancy, although the sensitivity is higher for renal and other upper urinary tract tumors than compared to lower tract tumors such as bladder cancer.16
Disposition
Symptomatic patients with a significant decline in their renal function, decreases in their hemoglobin or hematocrit, proteinuria, red blood cell casts, or muddy brown casts should prompt admission for further work-up. The stable patient who presents due to a limited number of episodes of hematuria, with stable renal function and blood counts, and who can void spontaneously can be discharged to home with outpatient urology referral. A similar patient found to have a UTI can be discharged home with oral antibiotics, but should be instructed to follow up with their primary care provider to ensure resolution of the hematuria after successful treatment of their UTI. In one study of over 4000 patients presenting for evaluation of hematuria, 18% of patients with gross hematuria were found to have malignancy.17 Therefore, if benign causes have been ruled out or treated, referral to urological specialist is needed for further imaging or cystoscopy to rule out malignancy.
TAKE-HOME POINTS
- Gross hematuria in the unstable patient can be a life-threatening cause of hemorrhagic shock.
- Urinary obstruction should be treated with urology consult and placement of a three-way catheter for decompression and continuous bladder irrigation (CBI). Watch out for post-obstructive diuresis.
- For imaging, CT urogram is appropriate for the otherwise stable patient with macroscopic hematuria—otherwise consider CT abdomen/pelvis with IV contrast.
- Ill patients or those with significant renal impairment or anemia require admission for further work-up.
- Patients otherwise appropriate for discharge should receive urology referral for follow-up as a high percentage of patients will go on to have malignancy.
| A Word Regarding Pediatrics A urinalysis is often the only laboratory test initially obtained during the evaluation of a child in the ED, but it can provide an important window into a child’s renal function. Children presenting with hypertension (above the 95th percentile for age), decreased urine output, and edema who are found to have proteinuria or cellular casts in combination with hematuria on a UA should receive a robust work-up including complete blood count, complete metabolic panel with renal profile, renal ultrasound, pediatric nephrology consult, and admission.18, 19 The differential diagnosis for such a presentation contains significant, life-threatening diseases: post-infectious glomerulonephritis, Henoch-Schonlein purpura (HSP), hemolytic-uremic syndrome (HUS), membranoproliferative glomerulonephritis, IgA nephropathy, and focal segmental glomerulosclerosis (FSGS). These conditions affect adults as well, but in the pediatric population where less laboratory evaluation is usually performed, an abnormal urinalysis in isolation could alter the entire diagnostic work-up in the right clinical context. However, an otherwise well-appearing child with normal vital signs with isolated microscopic hematuria should receive a referral to their pediatrician to have repeat urinalysis performed, similar to the recommendation from the AUA for adults.18 |
Case Resolution
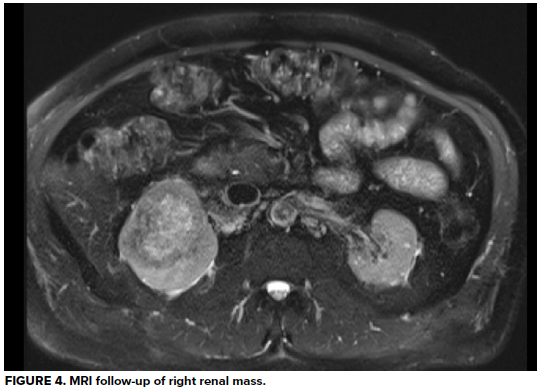
Urology is consulted; a 3-way Foley catheter is inserted, and continuous bladder irrigation is started. The patient is admitted for acute urinary obstruction and continued hematuria. He undergoes MRI for evaluation of the renal mass, which reveals 7.7cm right renal cell carcinoma, but fortunately no evidence of locally metastasis. Several days later he undergoes radical nephrectomy of the right kidney and pathology confirms diagnosis of clear cell carcinoma. Due to his CKD, he requires initiation of hemodialysis and receives several sessions before being discharged four days post-operatively. He continues on hemodialysis, grateful for his care, and has returned to managing his construction business.
References
- Davis R, Jones JS, Barocas DA, et al. Diagnosis, evaluation and follow-up of asymptomatic microhematuria (AMH) in adults: AUA guideline. Journal of Urology. 2012;188(6S):2473-2481. doi:10.1016/j.juro.2012.09.078
- Willis GC, Tewelde SZ. The approach to the patient with hematuria. Emergency Medicine Clinics of North America. 2019;37(4):755-769. doi:10.1016/j.emc.2019.07.011
- Mohr DN, Offord KP, Owen RA, Melton LJ. Asymptomatic Microhematuria and Urologic Disease: A Population-Based Study. 1986;256(2):224–229. doi:10.1001/jama.1986.03380020086028
- Germann CA, Homes, JA. Selected Urologic Disorders. In: Hockberger RS, Gausche-Hill M, Bakes K, et al. Rosen's Emergency Medicine Concepts and Clinical Practice. 9th Philadelphia, PA: Elsevier; 2018:1230-1231.
- Mark JR. Genitourinary Cancer. In: Porter RS, Kaplan JL, Lynn RB, et al. The Merck Manual. 20th Kenilworth, NJ: Merck Sharp & Dohme Corp; 2018:2100-2103.
- Gunn AJ, Patel AR, Rais-Bahrami S. Role of angio-embolization for renal cell carcinoma. Curr Urol Rep. 2018;19(10):76. doi:10.1007/s11934-018-0827-7
- Frey GT, Sella DM, Atwell TD. Image-guided renal intervention. Radiologic Clinics of North America. 2015;53(5):1005-1019. doi:10.1016/j.rcl.2015.05.002
- Rosenblatt L. Non-Traumatic Hematuria. In: Mattu A, Swadron S, ed. CorePendium. Burbank, CA: CorePendium, LLC; 2021. https://www.emrap.org/corependium/chapter/recPKai6P0ZJZnsOo/Non-Traumatic-Hematuria. Accessed September 26, 2021.
- Kotsikoris I, Papas TT, Papanas N, et al. Aortocaval fistula formation due to ruptured abdominal aortic aneurysms: a 12-year series. Vasc Endovascular Surg. 2012;46(1):26-29. doi:10.1177/1538574411418842
- Corbo J, Wang J. Kidney and ureteral stones. Emergency Medicine Clinics of North America. 2019;37(4):637-648. doi:10.1016/j.emc.2019.07.004
- Brisbane W, Bailey MR, Sorensen MD. An overview of kidney stone imaging techniques. Nat Rev Urol. 2016;13(11):654-662. doi:10.1038/nrurol.2016.154
- Abdominal and Pelvic Trauma. In: Stewart RM, Rotondo MF, Henry SM, et al. ATLS. 10th Ed. Chicago, IL: American College of Surgeons; 2018:88-92.
- Leslie SW, Sajjad H, Sharma S. Postobstructive Diuresis. In: StatPearls. Treasure Island, FL: StatPearls Publishing; 2021. URL: https://www.ncbi.nlm.nih.gov/books/NBK459387/. Accessed September 26, 2021.
- Hockberger RS, Schwartz B, Connor J. Hematuria induced by urethral catheterization. Annals of Emergency Medicine. 1987;16(5):550-552. doi:10.1016/S0196-0644(87)80683-2
- Wolfman DJ, Marko J, Nikolaidis P, et al. ACR Appropriateness Criteria® Available at https://acsearch.acr.org/docs/69490/Narrative/. American College of Radiology. Accessed 31 May 2021.
- O’Connor OJ, McSweeney SE, Maher MM. Imaging of hematuria. Radiol Clin North Am. 2008;46(1):113-132, vii. doi:10.1016/j.rcl.2008.01.007
- Edwards TJ, Dickinson AJ, Natale S, Gosling J, Mcgrath JS. A prospective analysis of the diagnostic yield resulting from the attendance of 4020 patients at a protocol-driven haematuria clinic. BJU Int. 2006;97(2):301-305. doi:10.1111/j.1464-410X.2006.05976.x
- Meyers KEC. Evaluation of hematuria in children. Urologic Clinics of North America. 2004;31(3):559-573. doi:10.1016/j.ucl.2004.04.015
- Dillman JR, Rigsby CK, Iyer RS, et al. ACR Appropriateness Criteria®Hematuria-Child. Available at https://acsearch.acr.org/docs/69440/Narrative/. American College of Radiology. Accessed 31 May 2021.