From unexplained syncopal events to undiagnosed palpitations, there are many cardiac conditions where patients benefit from closer monitoring.
Event monitors and Holter monitors have been traditionally used to observe cardiac rhythm in patients, but these devices are externally worn and cumbersome, limiting their use. Implantable loop recorders (ILRs), on the other hand, are inserted subcutaneously and are able to continuously record cardiac activity for up to 3 years.1
Unlike the more invasive implantable cardiac defibrillator, pacemaker, or biventricular device, insertion of ILRs is less invasive and is often placed in an outpatient setting under local anesthesia.
However, rare complications of infection or migration of the device can occur, and when these patients come to the emergency department (ED), the emergency physician must be able to discern which cardiac device the patient has implanted and just how life-threatening the presentation might be.
Case
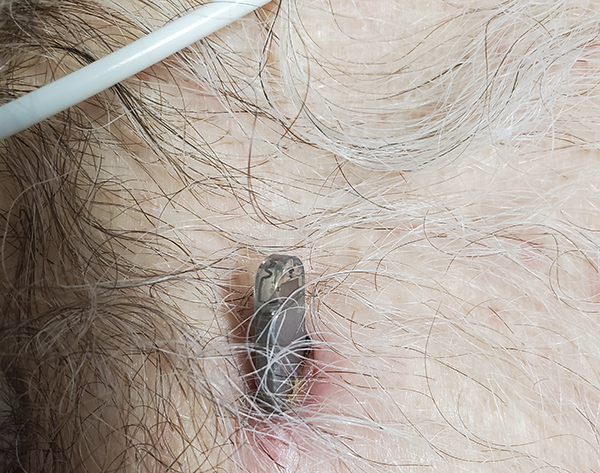
A 63-year-old male presented to the ED with a chief complaint of “my cardiac device is coming out of my skin.” The device had been implanted two weeks prior to monitor his palpitations. He reports he had woken up this morning and noticed the device sticking halfway out of his chest. Other than mild pain directly at the site of open skin, he denied any chest pain, palpitations, or dizziness. He denied any recent trauma to the chest, fevers, chills, erythema, purulent discharge, palpitations, shortness of breath, or diaphoresis.
The patient was afebrile and hemodynamically stable upon triage. Physical exam showed a small cardiac device protruding about 2 cm from the patient’s skin on his anterior chest wall. There was mild erythema and tenderness to palpation but no purulent discharge. His cardiac exam demonstrated a regular rate and rhythm, without any evidence of murmurs, rubs, or gallops. Additionally, his lung exam demonstrated bilateral, clear breath sounds.
EKG showed normal sinus rhythm. Chest x-ray showed clear lung fields and a normal-sized heart. The ILR was seen to the left of the sternum between the fourth and fifth ribs. There was no visible hematoma, tracking gas, or pneumothorax evident on imaging.
Upon further investigation, he was able to tell us that the device was an implantable loop recorder. The cardiac electrophysiologist team came to assess the patient and extracted the ILR. The 1.5 cm wound was left open to heal, and the patient was discharged home on an oral antibiotic for 7 days without any further complications.
Discussion
When a patient presents to the emergency department with an implanted cardiac device extruding from the skin, the first question an emergency physician must ask after ensuring that the patient is hemodynamically stable is: what kind of device is this? As the patient may not always know the answer, it is useful to generally be aware of the different types and placement locations of cardiac devices.
The most commonly encountered devices are implantable cardiac defibrillators, pacemakers, biventricular devices, and implantable loop recorders. Of these, implantable cardiac defibrillators, pacemakers, and biventricular devices all share a common insertion and placement site with a subcutaneous battery on the left side of the chest around the third intercostal space, wires traveling from the battery through the left subclavian vein, through the superior vena cava, and terminating inside the right ventricle (or the coronary sinus vein, in the case of the biventricular device). As opposed to the other devices, ILRs are minimally invasive – generally, a shallow incision is made at the left parasternal area at the level of the 4th–5th intercostals, the device is inserted subcutaneously, and the incision is closed with sutures, sterile strips, glue, or a combination.1,2 Unlike the other devices, ILRs are not in any way connected to the heart or vasculature and therefore pose minimal risk of complications – unless they migrate.
While complications of implantable loop recorders are rare, devices can be subject to migration and erosion through the skin into the cardiac or pleural space, incision site infection, and device malfunction.
- In a study of 154 patients, only 1 patient (0.6%) had device erosion through the skin, and none had site infections.2
- In another study of 133 patients, 2 patients (1.5%) had device erosion and 3 patients (2.3%) had site infections.3
- A study of 386 patients reported complications in 3.3% of patients, including infection, pain, and device malfunction - but without mention of device erosion through the skin.4
- A retrospective study in India noted an unusually high rate of device erosion through the skin, with as many as 4 out of 85 implants (4.7%) spontaneously self-extruding.5 These extrusions all occurred within 7-24 days of implantation, and it was postulated that the depth of the incision was the inciting factor.
A more worrisome case report documented the migration of an ILR through the pectoralis major muscle and into the left pleural space of the lungs.6 This patient presented with pleuritic chest pain, shortness of breath, and diaphoresis. A CT scan confirmed the displaced ILR, for which the patient required thoracoscopic surgery for removal (but ultimately recovered well).6
Though exceedingly rare and only seen in case reports, migration of the ILR into the deeper tissue – including the pleural space and the mediastinum – rather than erosion through the skin is a potentially lethal complication, the emergency physician must beware.
Management of Displaced ILRs
In a patient presenting with cardiac device complications, once the device has been successfully identified as an ILR rather than a more invasive device, the management is often straightforward.
Painful ILRs often require extraction. For those with suspected superimposed infection, a trial of antibiotics is often attempted first; if unsuccessful, the patient will require extraction of the ILR and a subsequent antibiotic course.
Self-extruding ILRs can simply be pulled or milked out, with hemostatic pressure applied to the site afterward. X-ray imaging is only necessary if it is thought there might be additional complications.
There are no guidelines on the management of the site after a self-extruding ILR has been removed, so it is left to the physician’s clinical judgment whether the patient requires wound closure or antibiotics. For the rarer and more dangerous cases of deeper ILR migration, further imaging (such as CT scan) and surgical intervention are required.
Key Points
- While implantable cardiac defibrillators, pacemakers, and biventricular devices are invasive and enter the right ventricle of the heart through the superior vena cava, implantable loop recorders are subcutaneous and not connected to any vasculature.
- Complications of ILRs include device migration and erosion through the skin, site infection, excessive site pain, device malfunction, and - rarely - migration into the pleural space.
- Removing an ILR that is eroding through the skin is as simple as milking the device out of the skin. No imaging is required prior to or after removal unless further complications are suspected.
- After removing the ILR, wound closure with glue, sterile strips, or sutures and prophylactic antibiotics are optional but may assist healing and help prevent future infections.
References and Recommended Reading
- Vilcant V, Kousa O, Hai O. Implantable Loop Recorder. [Updated 2020 Jul 31]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK470398/
- Maines, Massimiliano et al. Clinical impact, safety, and accuracy of the remotely monitored implantable loop recorder Medtronic Reveal LINQTM, EP Europace, Volume 20, Issue 6, June 2018, Pages 1050–1057, https://doi.org/10.1093/europace/eux187
- Bezzerides, Vassilios et al. The Real-World Utility of the LINQ Implantable Loop Recorder in Pediatric and Adult Congenital Heart Patients, JACC: Clinical Electrophysiology, Volume 5, Issue 2, 2019, Pages 245-251, https://doi.org/10.1016/j.jacep.2018.09.016.
- Ibrahim OA, Drew D, Hayes CJ, McIntyre W, Seifer CM, Hopman W, Glover B, Baranchuk AM. Implantable loop recorders in the real world: a study of two Canadian centers. J Interv Card Electrophysiol. 2017 Nov;50(2):179-185. doi: 10.1007/s10840-017-0294-y. Epub 2017 Nov 2. PMID: 29098486.
- Zakhar, Joseph et al. “Un-LINQed: Spontaneous extrusion of newer generation implantable loop recorders.” Indian pacing and electrophysiology journal vol. 20,5 (2020): 189-192. doi:10.1016/j.ipej.2020.04.005
- Preminger, Mark W et al. “Migration of an implantable loop recorder into the pleural space.” HeartRhythm case reports vol. 3,11 539-541. 3 Aug. 2017, doi:10.1016/j.hrcr.2017.07.012