Hypoglycemia in pediatric patients can be described as the lupus of metabolic disorders. It can be hard to differentiate if hypoglycemia is causing symptoms or is the result of an underlying problem.
Case 1. A 15-day-old male born full term with no prior hospitalizations or medical history is transferred to your pediatric emergency department from a small rural emergency department with the chief complaint of "lethargic since this morning." At the transferring facility the infant was responsive only to painful stimuli, was hypothermic, and had a partial sepsis workup done (everything except the LP); he received a 20 cc/kg bolus while awaiting EMS transport.
Case 2. A 2.5-day-old female born at the other hospital in town, full term with no complications, but who has not been latching well, arrives to your emergency department "looking blue" and is rushed back to a bed. While nurses are rushing to get vitals, you put the patient on a monitor and attempt to get a weight, if time allows. The neonate appears a little irritable but is consolable with mom. Vitals demonstrate an afebrile, tachycardic, and tachypneic baby. Your primary survey demonstrates the same, no murmurs, but you appreciate peripheral cyanosis with some perioral cyanosis. You ask to start a line, get labs, culture, UA and urine culture, place the patient on oxygen, and go to grab the ultrasound to do a bedside ECHO.
Case 3. A 4-year-old girl with no medical or surgical history arrives at your ED with mother who brought her in because she has been acting "funny" at home. On arrival you find a healthy- appearing girl sleeping in her mother's arms. When you attempt to wake her she wakes up to voice. Your primary survey was unimpressive. You ask the nurse to get vitals while you go to the computer to input orders. On your way over you hear the mother yelling, "She's having a seizure, help!" You quickly hurry back to the room and observe the girl having a generalized tonic-clonic seizure.
Background
Hypoglycemia in pediatric patients can be described as the lupus or syphilis of metabolic disorders. It can be hard to differentiate if the hypoglycemia is the cause of the symptoms or is a result of some underlying disorder like hyperinsulinism, glycogen storage disorders, fatty acid disorders, hormonal deficiencies and metabolic defects, or if it is a result of sepsis, cardiac and CNS abnormalities.
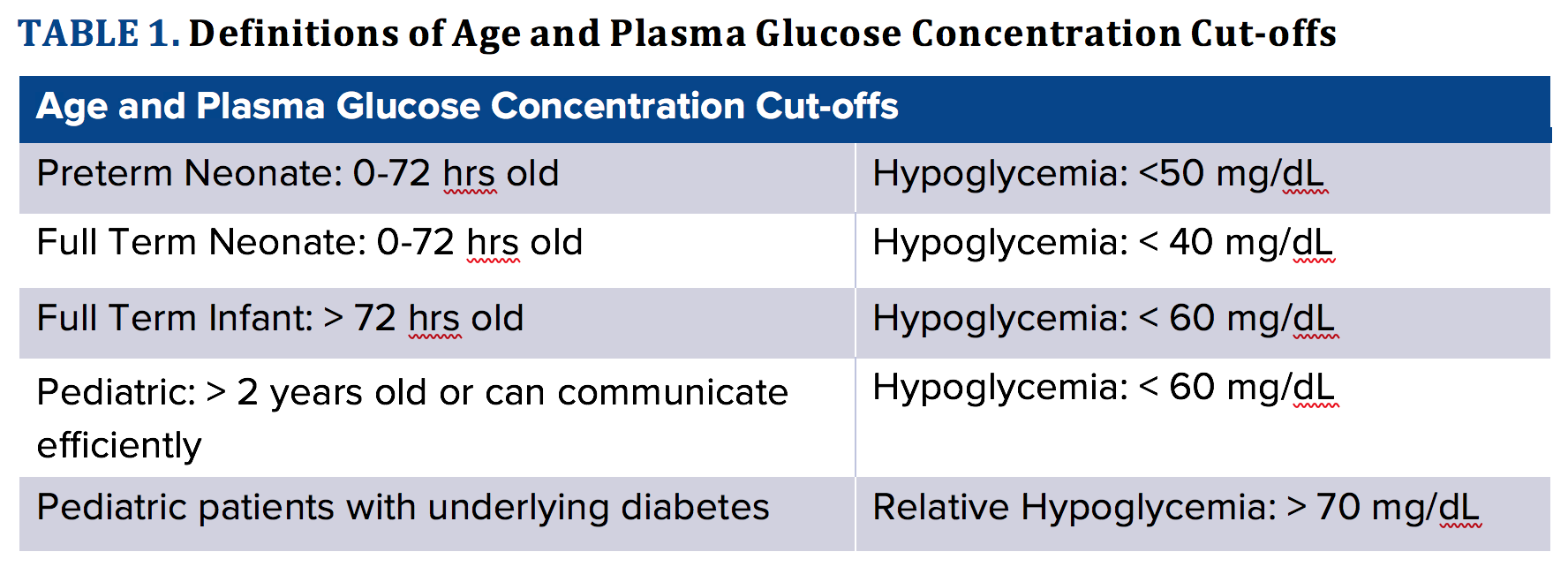
Hypoglycemia can be divided into asymptomatic and symptomatic. Asymptomatic hypoglycemia is defined at a preset plasma glucose (PG) level without any associated signs or symptoms of hypoglycemia. Symptomatic hypoglycemia is defined at a preset PG level with associated signs and symptoms of hypoglycemia. The cutoff PG value to define pediatric hypoglycemia varies based on hospital protocol and even among researchers. This debate is especially pertinent in the neonatal age group because neonates can have normally low glucose levels - as low as 35 mg/dL and remain asymptomatic - or as high as 45 mg/dL and present with symptoms of hypoglycemia. Furthermore, there is a debate as to the age cutoff that differentiates a neonate versus an infant. See Table 1 for the most common PG value cutoffs that, even when a patient is asymptomatic, should be treated as well as the most commonly used age cutoffs for neonates and infants. Diabetic patients, as they are used to higher levels of PG, can become symptomatic at higher PG levels. Making a strict definition that applies to all pediatric patients is difficult; therefore, we used what we understood to be widely accepted cutoffs.
The major energy source for the fetus, neonate, and pediatric patient is glucose. The pediatric patient's brain, especially in neonates and infants, utilizes up to 90% of the total body glucose. Additionally, pediatric patients have a higher rate of glucose turnover per kg because the
brain-to-body ratio is much higher than in adult patients. These characteristics combine to give pediatric patients an inherently higher risk of hypoglycemia. At birth there is an interruption of the constant glucose supply from the placenta, leaving the neonate to utilize their own body stores of glucose via breakdown of glycogen, free fatty acids, and protein. Neonatal glycogen stores are quickly depleted, and gluconeogenesis begins within hours of birth. At about
4-8 hours after birth, most neonates are at a steady state of PG levels, but will soon require an external source of glucose due to the real risk of hypoglycemia, which when prolonged can have long-term neurologic sequelae.
Signs and Symptoms
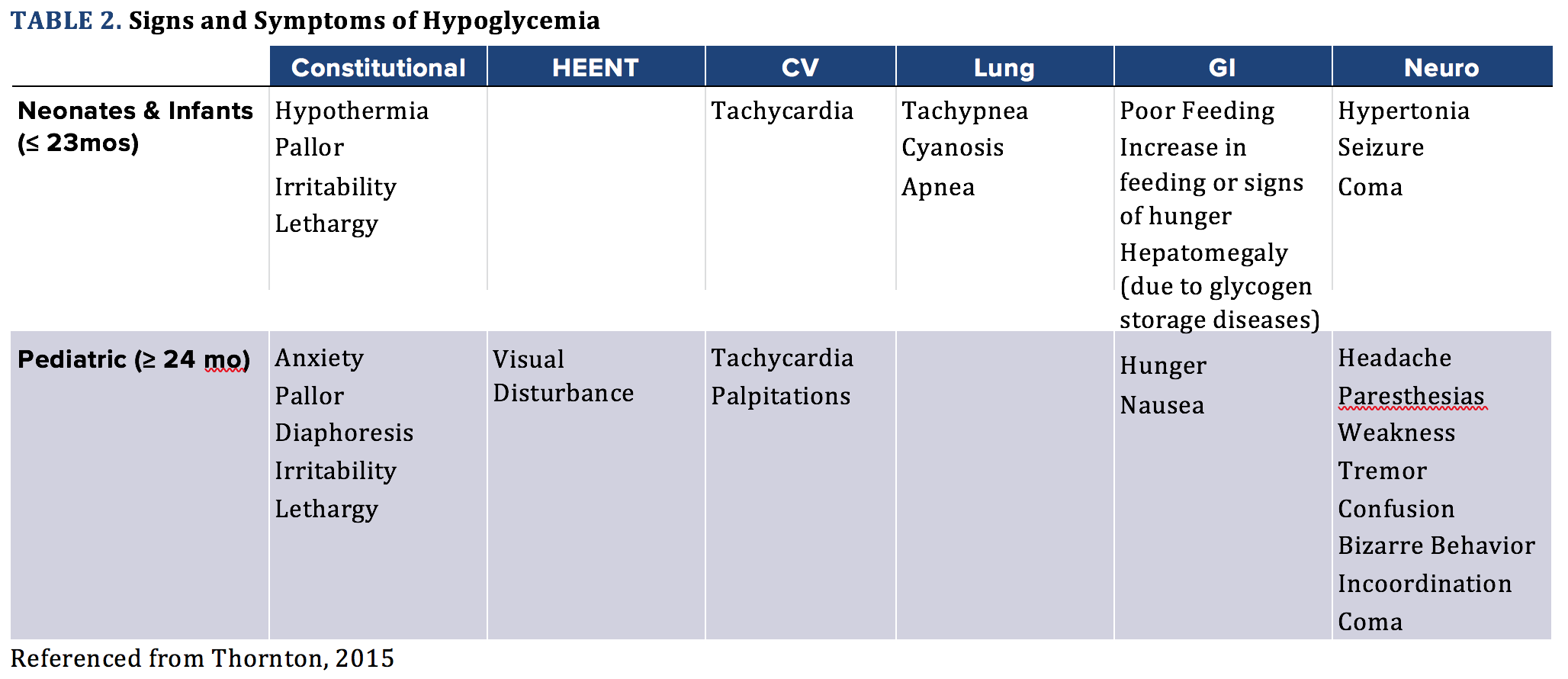
In adults or verbal children there can be more obvious signs of hypoglycemia like hunger or irritability. However, neonates, infants, and young pediatric patients are not able to verbalize their symptoms; therefore, the trained clinician must be aware of the subtle signs and symptoms. Hypoglycemia symptoms can be mistaken for toxidromes, postictal state, sepsis, meningitis, congenital cardiac abnormality, and many other diagnoses (see Table 2).
PEARL: When otherwise healthy patients present confused, with seizure-like activity, altered, and appear to have a toxidrome, always check their blood sugar!
Work-up
For the infant or child with hypoglycemia not easily explained by history (eg, a diabetic child who received insulin but failed to eat, or a clear-cut oral hypoglycemic ingestion), the emergency physician plays a vital diagnostic role. Historical facts to hone in on in all patients include symptom onset and progression, relationship with food/feeding. For neonates and young infants, birth weight and history should also be considered. Family history of any metabolic or genetic disorders should be elicited.
Physical exams should include any evidence of dysmorphic features, metabolic abnormalities, and toxidrome findings. For example, patients with hypothyroidism may have poor muscle tone, macroglossia, large fontanelles, and hoarse crying, while patients with hypopituitarism can have cleft palate, small for age/short stature, and/or micropenis. Patients with adrenal insufficiency can present with hyperpigmentation, anorexia, and weight loss. Glycogen storage disorders in patients may be hinted at with exam findings of hepatosplenomegaly.
Basic labs including urine analysis, CBC, BMP should be drawn, but if you get a POC glucose that is low, you should treat the hypoglycemia without waiting for a laboratory glucose. A plasma glucose is also important due to the small, but sometimes significant discrepancy in values between the lab glucose and the POC glucose. To identify the primary cause of the hypoglycemia, additional labs should be considered to evaluate for sepsis and metabolic disorders. This could include plasma insulin levels if there is a suspected hyperinsulinemia or insulin overdose, as well as free fatty acids and C-peptide levels. High plasma insulin levels with a low C-peptide level can indicate exogenous insulin exposure as endogenous insulin is attached to C-peptide prior to activation and would have similar C-peptide levels.
Even if you haven't identified a specific set of tests that would help you with diagnosis in the ED, it may be beneficial to the admitting team to have samples of urine and blood at the time of presentation and prior to administration of glucose to aid in diagnosis (Table 3).

Generally, imaging is not required in the ED once your suspicion for hypoglycemia is confirmed with a low PG. The exception to this rule is if you are considering a traumatic or neoplastic cause to the altered mental status or seizure that the patient may be experiencing.
Treatment
Asymptomatic Patients
Once hypoglycemia is confirmed via a low POC or plasma glucose level, treatment should be initiated regardless of patient symptoms. If the patient is fully conscious and is able to drink and/ or eat without risk of aspiration then a simple sugar either via glucose tablet/ gel, sugar, juice or similar should be given and a glucose rechecked in 10-20 minutes. Another attempt at oral glucose supplementation can be made or parenteral glucose should be considered if recheck continues to demonstrate hypoglycemia.
Symptomatic Patients
Similar to asymptomatic patients, the oral route is preferred for hypoglycemia treatment if the patient is conscious and can tolerate oral glucose administration. However, patients with the inability to protect their airway due to altered mental status, severe lethargy, or active seizures must receive immediate parenteral replacement with IV dextrose and/or IV/IM glucagon (see Table 4).
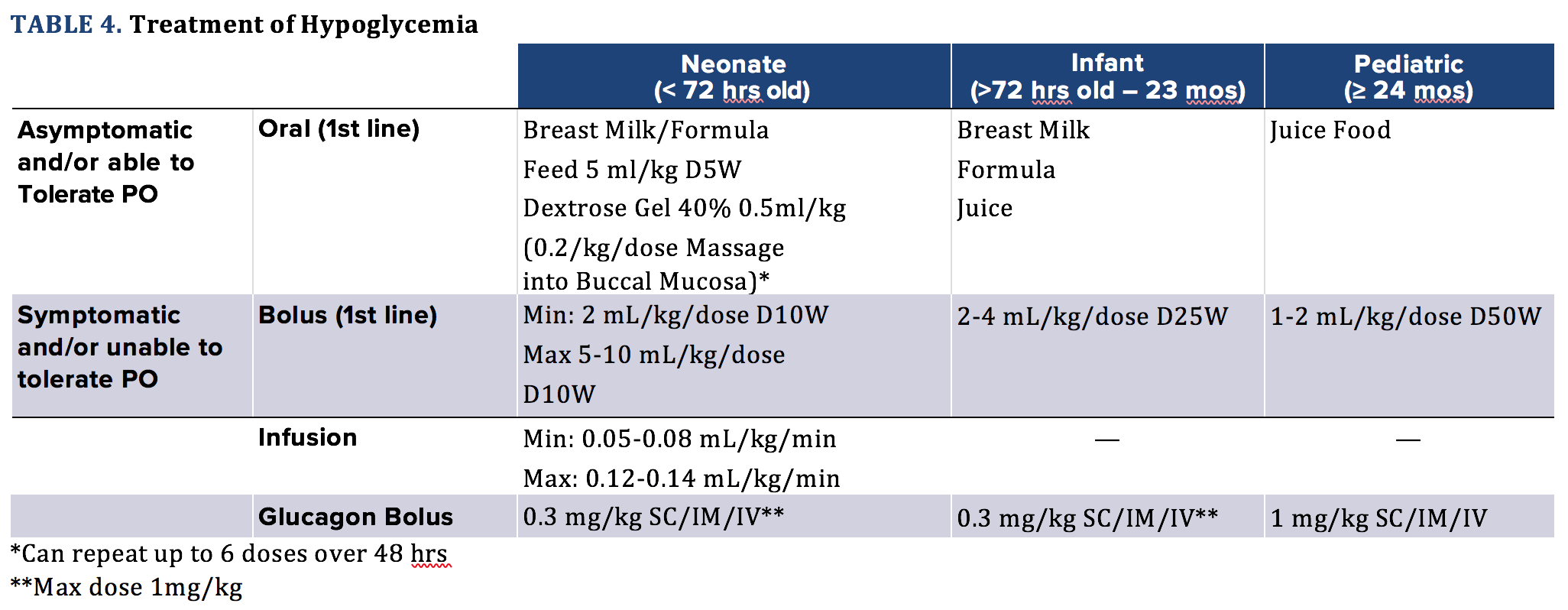
A bolus should be administered initially with frequent rechecks of glucose level. If the patient continues to be hypoglycemic in the setting of multiple boluses, the next step would be a continuous infusion of dextrose containing fluids. After starting an infusion of dextrose, PG levels should be monitored every 30-60 minutes or per your hospital nursing protocol.
Goal blood glucose is a concentration above 40 mg/dL, 50 mg/dL, or 60 mg/ dL in neonates, infants and pediatrics, respectively. Sulfonylurea ingestions are a special case of hypoglycemia and these patients should also be considered for administration of octreotide. Excessive insulin administration can be treated with glucagon in addition to IV boluses of dextrose. Do not use D25 or D50 or large volume boluses as they can create rebound hypoglycemia in hyperinsulinemic patients and can cause dangerous shifts in plasma osmolality. Furthermore, in high concentrations, particularly in neonates it may cause venous sclerosis and local tissue damage, partially due to the small diameter of neonate and infant veins. D10 is 10g of glucose in 100 mL water, D25 is 25g of glucose in 100 mL water, and so on.
(Referenced from Lexicom and Thornton)
Disposition
In general, if a patient appears to present as a pure hypoglycemic episode based on history, physical exam and diagnostic testing (if deemed necessary) short term observation for 1-2 hours, oral glucose replacement and glucose checking should be done and the patient can be safely discharged. However, if there is thought that the cause of hypoglycemia is due to ingestion, infection, metabolic abnormality, etc then one must be vigilant and admit the patient for observation even if history, physical exam and labs are not diagnostic for any serious pathology in the ED.
CASE 1 CONTINUED
The 15-day-old infant arrives to the ED via EMS, who state the patient was tachycardic, tachypneic, hypothermic at 95 F. The transferring facility only had a CBC by the time EMS arrived. As the patient is being placed on a monitor, you evaluate the ABCs and request a POC glucose. Patient is hemodynamically stable, but "punky" appearing on the primary survey. The nurse reports that the glucose is 38. Using the patient's weight, you request a 2 mL/kg bolus of D25W in addition to a rainbow blood draw for cultures and other "zebra" testing, urinalysis, urine culture and glucose recheck in 10 minutes. After administration of the glucose, infant appears more awake and interactive. Tachycardia and tachypnea are improved, and the repeat glucose is 57. Mom states she has had trouble with her son eating well on the breast and that he would go hours without a good feed. You ask the nurse to bolus another 2 mL/kg of D25W. After the second bolus the patient appears very well, is interactive and attempting to eat, with vital signs stabilized to normal. The recheck glucose is 84 and you tell mom she can feed the infant. After observation the infant's glucose stabilizes on several rechecks, all labs come back as normal, and the infant looks terrific. However, you still perform an LP, start IV antibiotics, and admit the patient to rule out sepsis, inborn error of metabolism, etc. You find out the patient is discharged in the next 48 hours as the blood cultures came back normal and
no other lab values were abnormal. The mother was educated to supplement formula and to not allow more than 2 hours to go by between meals with close PCP follow-up the next day.
CASE 2 CONTINUED
Bedside ultrasound appears abnormal with an overriding aorta and possible VSD. The technician, who decided to check a POC glucose tells you that the glucose is 34, which prompts you to have the nurse bolus 2 mL/kg of D10W. Soon after the bolus the baby appears to perk up but continues to be cyanotic. Repeat POC glucose is 45. While waiting for labs to return you bolus the neonate with 10/ cc/kg of ½ NS with D10 as well as IV antibiotics, 20 cc/kg NS, and then call PICU and cardiology. You admit the patient for continued work-up with a broad differential including congenital heart disease, sepsis, and inborn errors of metabolism.
CASE 3 CONTINUED
The seizure is short lasting and per the mom the girl has no history of seizures. You ask the nurse to check a glucose, which you find to be 32, so you have the nurse attempt to get IV access to allow you to bolus the patient with 1 mL/ kg of D50W. The nurse is unsuccessful
at the first IV attempt, so while getting the other IV you have another nurse give a 0.3 mg/kg IM shot of glucagon, while you get the IO kit ready if the nurse is unsuccessful on her second attempt. The nurse gets the IV and you ask her to draw a rainbow of labs and to bolus with D50W. On reassessment the patient appears lethargic now and is only responding to painful stimuli. You recheck the glucose and it is 34. Nothing on the history you got from mom was concerning besides the acute onset of symptoms today after mom picked the patient up from grandma's house. You have mom call grandma and find out that some of her "sugar pills" are missing, but she doesn't know which one as it's missing from her weekly pill box; she "thinks the medicine starts with a 'G'." Sulfonylureas are on your differential, so you also give a bolus of octreotide and place the patient on an infusion of glucose and admit to PICU.
Pearls for Evaluation, Treatment, and Disposition of Hypoglycemic Pediatric Patients
- Work-up
- Get a POC and plasma Glucose
- Consider CBC, CMP, lactate, ammonia, UA if not simple hypoglycemia
- Zebra labs should be drawn in conversation with Pediatric team, also think of obtaining a critical sample of labs and urine prior to treatment
- Oral Glucose
- Asymptomatic
- Tolerate PO
- Symptomatic
- 1st line is Parenteral Bolus
- Dextrose Rule of 50
- D10/D25/D50 @ rates of 5/2/1 mL/kg IV
- Disposition
- Monitor glucose levels 1-2 hrs or until patient is alert and can tolerate PO, then can discharge home with close PCP follow-up/ good return precautions
- If not, then admission recommended
- Neonates and infants: recommend admission for monitoring
References
1. Adamkin DH for the Committee on Fetus and Newborn. Postnatal glucose homeostasis in late-preterm and term infants. Pediatrics. 2011;127(3):575-579.
2. Balijepalli C, Druyts E, Siliman G, Joffres M, Thorlund K, Mills EJ. Hypoglycemia: a review of definitions used in clinical trials evaluating antihyperglycemic drugs for diabetes. Clin Epidemiol. 2017;9:291–296.
3. Bier DM, Leake RD, Haymond MW, Arnold KJ, Gruenke LD, Sperling MA, Kipnis DM. Measurement of “true” glucose production rates in infancy and childhood with 6, 6-dideuteroglucose. Diabetes. 1977;26(11):1016-1023.
4. Cornblath M, Schwartz R. Disorders of Carbohydrate Metabolism in Infancy. Philadelphia, PA: WB Saunders; 1966.
5. Gandhi K. Approach to hypoglycemia in infants and children. Transl Pediatr. 2017;6(4):408–420.
6. Gregory JW, Aynsley-Green A. Hypoglycaemia in the infant and child. Baillieres Clin Endocrinol Metab. 1993;7(3):683-704.
7. Haymond MW, Howard C, Ben-Galim E, DeVivo DC. Effects of ketosis on glucose flux in children and adults. Am J Physiol. 1983;245(4):E373-E378.
8. Reid SR, Losek JD. Hypoglycemia in infants and children. Relias Media. 2000;5(3):23.
9. Lexicomp Online. Glucagon: Pediatric Drug Information, Hudson, Ohio: Wolters Kluwer Clinical Drug Information, Inc.; 2013; April 15, 2013.
10. Sunehag A, Ewald U, Gustafsson J. Extremely preterm infants (< 28 weeks) are capable of gluconeogenesis from glycerol on their first day of life. Pediatr Res. 1996;40(4):553-557.
11. Thornton PS, Stanley CA, De Leon DD, et al. Recommendations from the pediatric endocrine society for evaluation and management of persistent hypoglycemia in neonates, infants, and children. J Pediatr. 2015;167(2):238-245.



