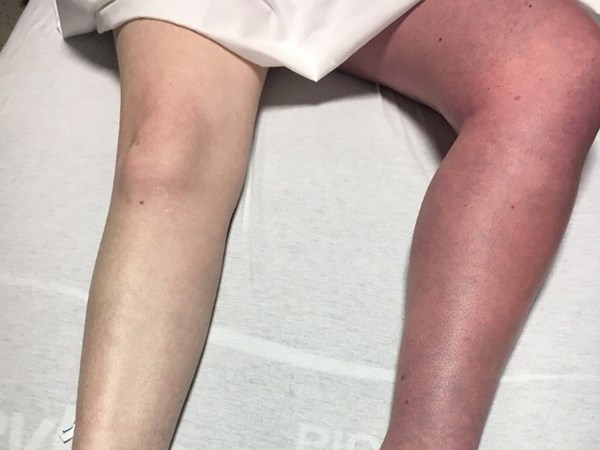
A 44-year-old female presents to the emergency department with acute left leg pain one hour prior to arrival.
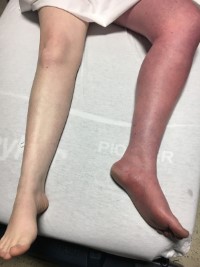
The patient states that she was bending over and felt a pop in her left leg. On exam, the left leg is discolored (Fig. 1), warm, swollen, and exquisitely tender to palpation. There is decreased sensation distal to the left knee. 1+ dorsalis pedis artery and posterior tibialis artery pulses are palpated on the left, 2+ on the right. A point-of-care ultrasound (POCUS) was performed (Fig. 2, Fig. 3, Fig. 4), which confirmed the diagnosis.
Question: What is the next best step in management from the ED?
- IV antibiotics and hospital admission
- Systemic fibrinolytics
- Discharge with oral antibiotics
- Urgent vascular imaging
 |
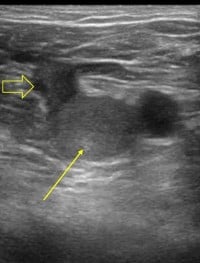 |
| Figure 1. Lower extremity physical exam findings | Figure 2. POCUS showing non-compressible, echogenic, occlusive thrombus at the junction of the left femoral vein (solid arrow) and left greater saphenous vein (open arrow). |
 |
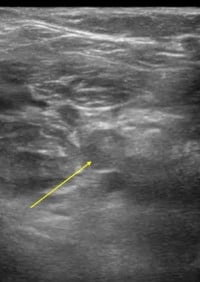 |
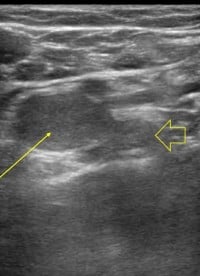
| Figure 3. POCUS showing non-compressible, echogenic, occlusive thrombus at the junction of the left femoral vein (solid arrow) and left deep femoral vein (open arrow). | Figure 4. POCUS showing non-compressible, echogenic, occlusive thrombus within the left popliteal vein (solid arrow). |
Answer
The patient in the clinical vignette is suspicious for phlegmasia cerulea dolens (PCD). The correct answer is D: urgent vascular imaging. Point-of-care ultrasound (POCUS) of the left leg (Fig. 2 – Fig. 4) demonstrated a non-compressible thrombus extending from the left common femoral region to below the left popliteal region. Vascular surgery was consulted, and the patient was promptly taken to the interventional radiology suite for concurrent mechanical and chemical thrombolysis.
Definition
PCD is a rare but emergent condition of massive venous thrombosis that can rapidly produce irreversible vascular gangrene.1,3 First described by Gregoire in 1934, PCD is recognized by the classic triad of extremity pain, swelling, and blue discoloration secondary to venous occlusion.1 Most cases of PCD are due to a DVT of an iliac or femoral vein4, and only 2-5% of PCD cases are caused by an upper extremity DVT.5 This blue discoloration differentiates PCD from phlegmasia alba dolens, which presents as a pale limb due to arterial spasm and may precede PCD.
Staging
In general, acute limb ischemia is classified by stages I-III: I (Viable), IIa (Marginally Threatened), IIb (Immediately Threatened), and III (Irreversible). Stage I (Viable) has no sensory loss, no muscle weakness, and audible doppler signals of the artery and vein. Stage IIa (Marginally Threatened) has sensory loss of the toes or no sensory loss, no muscle weakness, sometimes inaudible doppler signal of the artery, and audible doppler signal of the vein; the limb is salvageable with treatment. Stage IIb (Immediately Threatened) has sensory loss of more than the toes, mild to moderate muscle weakness, inaudible doppler signal of the artery, and audible doppler signal of the vein; the limb is salvageable with immediate revascularization. Stage III (Irreversible) has a profound sensory loss, muscle paralysis, and inaudible doppler signals of the artery and vein; this often requires amputation.6
Risk Factors
Risk factors for PCD include malignancy, hypercoagulable disorders, venous stasis, oral contraceptives, and a history of previous DVT. In a systematic review, over one-third of patients with PCD had a malignancy. In contrast, two-thirds of patients with PCD had a history of hypercoagulable disorder, venous stasis, oral contraceptive use, or previous DVT. However, in 16% of patients, there was no history of disease (n=62).1
Diagnosis
PCD diagnosis can be accomplished clinically with visualization of a swollen and cyanotic limb combined with positive DVT in the iliac or femoral veins confirmed by duplex ultrasonography,2 which uses both B-mode and doppler features. Contrast venography is considered the gold standard, but is less commonly utilized due to ultrasound's comparative safety, accessibility, and portability.
Complications
The major complications of PCD include pulmonary embolism, myonephrotic metabolic syndrome, and compartment syndrome.1
Pulmonary Embolism (PE): In patients with PCD, 29% had PE, resulting in a 50% mortality rate.1 PE is a life-threatening complication for 50% of untreated DVTs and contributes to 10-15% of all hospital deaths.7 All patients suspected of having a PE should be on telemetry, have frequent blood pressure monitoring and maintain oxygen saturation greater than 95%. IV crystalloid fluids are given to increase preload. These patients should be given therapeutic dosing of unfractionated heparin (preferred for renal insufficiency or increased subcutaneous tissue thickness) or low molecular weight heparin. Unfractionated heparin is given as an initial bolus of 80 units per kilogram, then 18 units per kilogram per hour with a goal activated partial thromboplastin time between 55 and 80 seconds.4 Depending on the disease burden, catheter directed thrombolytics or systemic thrombolytics may be indicated.
Myonephrotic metabolic syndrome (MNMS): MNMS is a type of ischemia-reperfusion injury caused by metabolites of muscle cells that have undergone liquefactive necrosis. When the clot is destroyed, reperfusion occurs, and the accumulated metabolites circulate in the body and can cause fatal hyperkalemia, pulmonary edema, metabolic acidosis, and myoglobinuria. MNMS can be prevented with aggressive fluid resuscitation and, if severe, hemodialysis.6
Compartment Syndrome: Similar to MNMS, compartment syndrome is caused by ischemic reperfusion leading to increased capillary permeability. The increased permeability results in edema which is contained in a compartment of the limb that is surrounded by inflexible fascia. A compartment pressure greater than 30mmHg may necessitate a fasciotomy to avoid permanent damage to arteries, veins, and nerves in the area.6
Treatment & Disposition
To treat PCD, immediate steps include neutral positioning of the limb, removal of constrictive clothing or dressings, fluid resuscitation with crystalloid fluids, initiation of anticoagulation, and immediate consultation for thrombolysis or thrombectomy from vascular surgery, interventional radiology, or peripheral interventional cardiology. If none of these specialists are available, consider 50 to 100 mg intravenous alteplase (tPA) infused over four hours, keeping the contraindications of intravenous thrombolysis in consideration. Of note, there is limited data to support a particular alteplase dosing regimen.4
Acute limb ischemia stages I (Viable) and IIa (Marginally Threatened) are initially treated endovascularly due to less ischemic burden and less time constraint, while stage IIb (Immediately Threatened) is treated with immediate surgical thromboembolectomy.6 Although many treatment options exist, urgent revascularization is universally required to prevent irreversible ischemia, amputation, and possibly death from cardiovascular collapse.1-3 Regardless of the revascularization method, all patients require DVT prophylaxis and close follow-up to elucidate inciting factors.1,3 Importantly, any patient undergoing thrombolytic therapy should be admitted to an intensive care unit.4
Case Conclusion
After the patient underwent concurrent mechanical thrombectomy and thrombolysis, an inferior vena cava filter was placed and she was discharged with anticoagulation. Of note, she had no known risk factors, was not pregnant nor on oral contraceptives, and had no prior thrombotic events. Her one-month follow-up appointment for further hypercoagulability workup was negative.
TAKE-HOME POINTS
- Identification: If your patient has a swollen and cyanotic limb, use duplex ultrasound to look for a DVT in the iliac or femoral veins. Always monitor peripheral pulses.
- Initial treatment: Put the affected leg in a neutral position, remove constrictive clothing or dressings, give intravenous crystalloid fluids, initiate anticoagulation, and immediately consult an interventional vascular team for thrombolysis or thrombectomy. If none of these specialists are available, consider administration of alteplase (tPA).
- Complications: Look out for pulmonary embolism, myonephrotic metabolic syndrome, and resulting arrhythmias, or compartment syndrome.
References
- Chinsakchai K, Ten Duis K, Moll FL, de Borst GJ. Trends in management of phlegmasia cerulea dolens. Vasc Endovascular Surg. 2011 Jan;45(1):5-14.
- Yang SS, Yun WS. Surgical Thrombectomy for Phlegmasia Cerulea Dolens. Vascular Specialist International. 2016; 32: 201-204.
- Suwanabol PA, Tefera G, & Schwarze ML. Syndromes Associated with the Deep Veins: Phlegmasia Cerulea Dolens, May-Thurner Syndrome, and Nutcracker Syndrome. Perspectives in Vascular Surgery and Endovascular Therapy. 2010; 22(4): 223–230.
- Tintinalli, Ma, Yealy, Meckler, Stapczynski, Cline, and Thomas. A Comprehensive Study Guide, 9th edition. 2019; Chapter 25: Thromboembolism, p 150, 154-155.
- Sullivan VV, Wolk SW, Lampman RM, Prager RL. Upper extremity venous gangrene following coronary artery bypass: a case report and review of the literature. Journal of Cardiovascular Surgery. 2001; 42(4), 551.
- Obara H, Matsubara K, Kitagawa Y. Acute Limb Ischemia. Ann Vasc Dis. 2018; 11(4): 443-448.
- Veltchev LM, Kalniev MA, Todorov TA. Phlegmasia cerulean dolens- risk factors and prevention. https://www.journal-imab-bg.org/statii-09/vol09_1_89-91str.pdf J IMAB. 2009;15:89–91.
- Kou CJ, Batzlaff C, Bezzant ML, Sjulin T. Phlegmasia Cerulea Dolens: A Life-Threatening Manifestation of Deep Vein Thrombosis. Cureus. 2020; Jun 12;12(6):e8587.