Supplemental oxygen is the most common intervention used when managing hypoxic patients and can be easily applied via nasal cannula connected to oxygen from the wall.
At rest, the inspired flow of a normal respiratory cycle is approximately 30 liters (L) per minute. When a patient is in respiratory distress, the inspired flow demand can exceed 100L/min. A traditional low-flow nasal cannula provides up to 6L/min of flow (though higher rates are possible), which is far from the demand of patients in respiratory distress. Although oxygen directly inside the tubing from the wall has a fraction of inspired oxygen (FiO2) of 100%, this mixes with ambient air and dilutes the amount of FiO2 the patient actually receives. For example, the FiO2 of a patient receiving 6L via nasal cannula is diluted to approximately FiO2 45%.1 This is one of the main limitations of low-flow nasal cannula; its low-flow state allows for mixing of ambient air with the oxygen source, ultimately resulting in a lower FiO2 delivered to the patient. Additionally, this system blows cool, dry air that causes nasal mucosal irritation at higher flow rates, which can lead to epistaxis. As mentioned above, higher flow rates are possible with a standard nasal cannula, but often are not well-tolerated by the patient because of discomfort.
In contrast, heated high-flow nasal cannula (HFNC) is capable of delivering 100% humidified and heated oxygen up to flow rates of 60L/min. This higher flow allows for higher amounts of concentrated FiO2 to be delivered and limits the amount of ambient air mixing in with the oxygen delivered. HFNC additionally may offer a small amount of positive end-expiratory pressure (PEEP) at high flow rates. As flow is increased on the device, this causes an increase in nasopharyngeal pressure. In general, every 10L/min increase of flow yields roughly 0.7 cm H2O of airway pressure when the mouth is closed and 0.35 cm H2O when the mouth is open.2 Additionally, HFNC offers the benefit of washout of carbon dioxide (CO2) from anatomical dead space.6 This decreases rebreathing of the expired CO2 present in the dead space, which can help with decreasing respiratory rate of the patient.
HFNC Setup
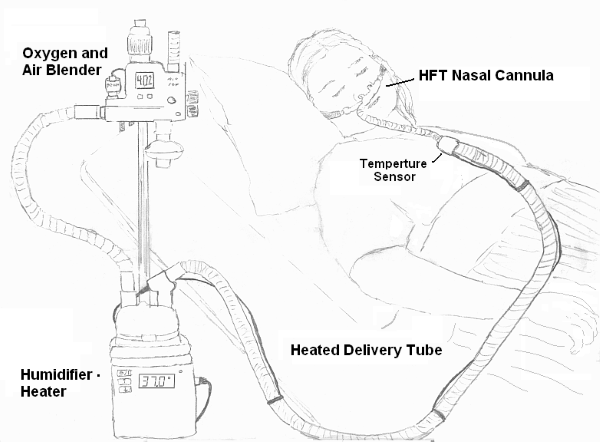
Figure 1: General setup of Heated High Flow Nasal Cannula (Wikimedia: Strangecow, 2011)3
The apparatus comprises an active heated humidifier, an air and oxygen blender, a single circuit, and a 2-prong nasal cannula.
The air and oxygen blender can set an inspiratory fraction of oxygen (FiO2) from 21% up to 100% in a flow up to 60 L/min.
The gas is heated and humidified and delivered through the circuit as shown in Figure 1.
 Figure 2: Example of a commonly used heated high-flow nasal cannula. Courtesy of Vapotherm.6
Figure 2: Example of a commonly used heated high-flow nasal cannula. Courtesy of Vapotherm.6
How to Titrate Heated HFNC
- First set the flow rate. Start at 20-35 L/min and titrate up as tolerated for hypoxia or patient comfort.
- Next, set the FiO2 and titrate to the desired peripheral oxygen saturation according to your pulse oximetry or PaO2.
- The flow rate can be increased in 5-10 L/min increments if breathing remains labored or oxygenation fails to adequately improve.
- Although increasing both flow rate and FiO2 will result in improved oxygenation, it is preferred to maximize the flow rate first in an effort to keep the FiO2 <60% when possible.
- Remember to re-assess your patient often for any signs of deterioration. These patients already have a significant oxygen requirement and may ultimately require intubation.
- The ROX Index available on MDCalc is a useful tool that can predict the need for intubation for patients on HFNC. This takes into account the patient's current pulse oximetry, current FiO2 requirement on the HFNC, and respiratory rate.
- A ROX index of >4.88 measured at 2, 6, or 12 hrs after initiation of HFNC is associated with lower risk for intubation. A ROX index of <3.85 suggests a risk that HFNC will fail and the patient will require intubation. An indeterminate value in between 3.85-4.88 may be repeated 1-2 hrs later for re-evaluation.
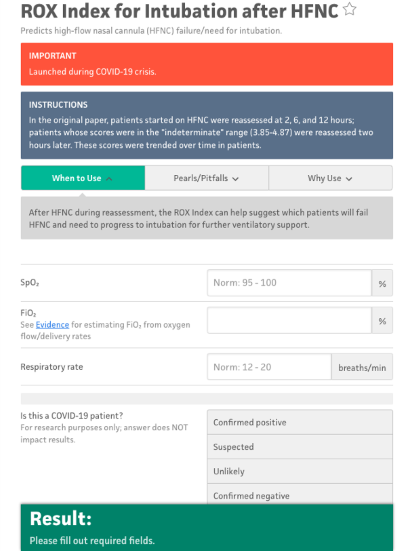
Figure 3: ROX Index, MDCalc7
Patient Populations Who May Benefit from HFNC
- Patients with hypoxemic respiratory failure secondary to dead space pathology (eg, pulmonary embolism)
- Patients with pre-load sensitive pathologies requiring high oxygen support who cannot tolerate large amounts of positive pressure (eg, right heart failure)
- May be used for preoxygenation prior to endotracheal intubation
- May be used as a non-invasive way to increase body temperature in patients with significant hypothermia6
Evidence for Use
FLORALI Trial4
A small, multicenter randomized open label study of patients with non-hypercarbic hypoxemic respiratory failure (P/F ratio <300) were randomized to HFNC, non-invasive positive pressure support (NIV), or conventional oxygen support via face mask. This study looked to compare rates of intubation in the first 28 days as well as mortality in the ICU, mortality at 90 days, and ventilator free days within the first 28 days. The primary outcome did not differ between groups; however, secondary outcomes favored HFNC.
- Ventilator-free days at day 28
- HFNC 24±8 days vs standard oxygen 22±10 (p=0.02)
- HFNC 24±8 days vs NIV 19±12 (p=0.02)
- Mortality at 90 days–hazard ratio 95% confidence interval
- HFNC vs standard oxygen 2.01 (1.01 to 3.99, p=0.046)
- HFNC vs NIV 2.50 (1.31 to 4.78, p=0.006)
Those receiving NIV often did not receive this intervention for a majority of the day and spent the other portion on face mask oxygen. This makes the comparison between HFNC and NIV more difficult, but possibly this reflects real world practice. An additional pitfall of this study is that the majority (>80%) of the patients studied had pneumonia, making it difficult to generalize these results to sepsis and other causes of hypoxemic respiratory failure without hypercapnia.
Pitfalls
- Cannot be used in patients with extensive trauma or abnormalities of the face, nose, or airway that would preclude being able to use an appropriate-fitting nasal cannula
- Has no direct effect in controlling ventilation of the patient
- Can provide a small amount of PEEP, although the exact level of PEEP the patient is receiving is difficult to reliably measure
- Therefore, it would not be as helpful in patients with hypoxemia requiring positive pressure (eg, cardiogenic pulmonary edema)
- Can take up to 10-15 min to set up the circuit in a patient who is critically ill with hypoxia
- Traditional HFNC requires oxygen directly to the wall to support its high flow, therefore making it impossible to transport patients who require HFNC. NOTE: The new HVT 2.0 by Vapotherm has an internal battery and rolls stand that allows for up to 2 oxygen cylinders to move a patient within the hospital for up to an hour.
References
- Sharma S, Danckers M, Sanghavi D, Chakraborty RK. High-flow nasal cannula. In: StatPearls. StatPearls Publishing; 2023.
- Drake MG. High-flow nasal cannula oxygen in adults: an evidence-based assessment. Ann Am Thorac Soc. 2018;15(2):145-155.
- Wikimedia Commons. Strangecow 2011. Offered for free use into the public domain. Accessed June 5, 2023.
- Frat JP, Thille AW, Mercat A, et al., for the FLORALI Study Group and REVA Network. High-flow oxygen through nasal cannula in acute hypoxemic respiratory failure. N Engl J Med. 2015;372(23):2185-2196.
- Gilardi E, Petrucci M, Sabia L, et al. High-flow nasal cannula for body rewarming in hypothermia. Crit Care. 2020;24(1):122.
- Image of Vapotherm Apparatus. Vapotherm. https://vapotherm.com/precision-flow/.
- Roca O. ROX Index for Intubation after HFNC. MDCalc. Accessed June 20, 2023.